|
상세 정보 |
|||
| 기술: | 염소 처리 - 화학적 소독 | 디자인: | 미끄럼 탑재 |
|---|---|---|---|
| 물 임시.: | 5~35˚C | ClO2 집중: | 0.2 2.5 mg/L에 |
| 물 유량: | 최고 100 m3/h | 시약 1: | 뿌로진 (3.36%에 있는 NaClO2) |
| 시약 2: | 하크들 15% | 보증: | 1년이요 |
| 재료: | SS304/SS316L,carbon 강철 | 전압: | 380V,415V,220V, 맞춤화될 수 있습니다 |
| 애플리케이션: | 물 정화 동작, 정수기, 제약의, 식수, 식량 | 사이즈: | 더 작거나 더 큰 1750*550*1650mm |
| 제어: | 전자적, 설명서 / PLC | ||
| 강조하다: | 2.5 mg/L 이산화 염소 가스 생생기,OEM 이산화 염소 가스 생생기,2.5 mg/L 이산화 염소 투약 단위 |
||
제품 설명
Chemical chlorine dioxide generator water treatment disinfection dosing device
The working principle and composition of chemical method
HB series chlorine dioxide generator uses two chemical reaction methods to produce chlorine dioxide.
- 2 dosing pumps in a
- 1 reaction vessel (optional)
- 1 redox probe (optional)
- 1 pump cabinet
- 1 control cabinet
- 1 Pulse flowmeter (optional)
- 1 static mixer
- Chemical drums
- Secondary containers
![]()
1. Sodium chlorate produces chlorine dioxide. The chemical reaction formula is as follows:
Main reaction: NaCLO3+2HCL↑CLO2+1/2CL2+NaCL+H2O
Side reaction: NaCLO3+6HCL↑3CL2+NaCL+3H2O
2. Sodium chlorite produces chlorine dioxide, the chemical reaction formula is as follows:
5NaCLO2+4HCL↑4CLO2+5NaCL+2H2O
The raw materials for the above reactions are accurately metered by their respective metering devices and then fed into the reactor.
The reaction generates chlorine dioxide gas, which is sucked by the jet and mixed with water to make an efficient chlorine dioxide disinfectant.
Features
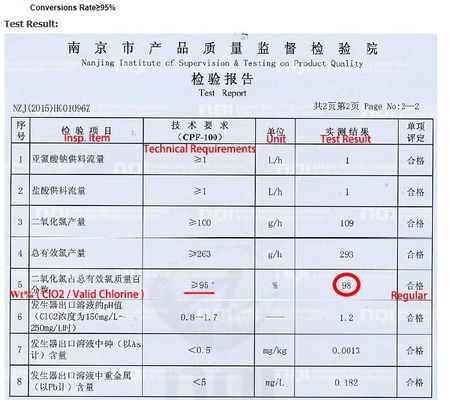
1. Adopt advanced raw material formula, high conversion efficiency, sodium cyanate above 70%, sodium chlorite above 95%.
2. Compact design structure, small floor space, simple operation, simple and safe maintenance, and long service life.
3. The degree of automation is high. The control system can automatically adjust the dosage according to the change of water quality, and is equipped with various safety protection measures such as water shortage and no medicine shutdown.
4. Complete specifications and varieties, chlorine dioxide generator from 50g-20000g/h.
| Model | Available chlorine volume(g/h) |
Weight (kg) |
Equipment Size(mm) |
Power water |
Power (kg) |
|
| Pipe diameter(mm) |
Pressure (Mpa) |
|||||
| CDG-100 | 100 | 70 | 480x450x1000 | 25 | ≥0.25 | 0.5 |
| CDG-200 | 200 | 72 | 500X450X1050 | 25 | ≥0.25 | 0.5 |
| CDG-500 | 500 | 85 | 550X500X1100 | 25 | ≥0.25 | 0.5 |
| CDG-1000 | 1000 | 96 | 600X550X1150 | 32 | ≥0.25 | 0.7 |
| CDG-3000 | 3000 | 129 | 650X600X1250 | 32 | ≥0.3 | 1.5 |
| CDG-4000 | 4000 | 147 | 700X650X1400 | 50 | ≥0.4 | 2.5 |
| CDG-5000 | 5000 | 165 | 750X700X1600 | 50 | ≥0.4 | 3.0 |
| CDG-7000 | 7000 | 182 | 800X750X1700 | 75 | ≥0.4 | 3.4 |
| CDG-20000 | 20000 | 265 | 1000X850X1900 | 90 | ≥0.4 |
4.5 |
1. What is stabilised Chlorine dioxide?
Like ozone and chlorine, chlorine dioxide is an oxidizing biocide and not a metabolic toxin. This means that chlorine dioxide kills microorganisms by disruption of the transport of nutrients across the cell wall, not by disruption of a metabolic process.
Stabilised chlorine dioxide is ClO2 buffered in an aqueous solution. Adding an acid to the required concentration activates the disinfectant.
Back to top
2. How does it work?
Of the oxidizing biocides, chlorine dioxide is the most selective oxidant. Both ozone and chlorine are much more reactive than chlorine dioxide, and they will be consumed by most organic compounds. Chlorine dioxide however, reacts only with reduced sulphur compounds, secondary and tertiary amines, and some other highly reduced and reactive organics. This allows much lower dosages of chlorine dioxide to achieve a more stable residual than either chlorine or ozone. Chlorine dioxide, generated properly (all chlorine dioxide is not created equal), can be effectively used in much higher organic loading than either ozone or chlorine because of its selectivity.
Back to top
3. How effective is it?
The effectivety of chlorine dioxide is at least as high as chlorines, though at lower concentrations. But there are more and important advantages.
The bactericidal efficiency is relatively unaffected by pH values between 4 and 10;
Chlorine dioxide is clearly superior to chlorine in the destruction of spores, bacteria's, viruses and other pathogen organisms on an equal residual base;
The required contact time for ClO2 is lower;
Chlorine dioxide has better solubility;
No corrosion associated with high chlorine concentrations. Reduces long term maintenance costs;
Chlorine dioxide does not react with NH3 or NH4+;
It destroys THM precursors and increases coagulation;
ClO2 destroys phenols and has no distinct smell;
It is better at removing iron and magnesia compounds than chlorine, especially complex bounds;